Solvent extraction of zirconium and hafnium is one of the common methods for separating zirconium and hafnium. Compared with other zirconium and hafnium separation methods (such as pyrolysis separation and solvent extraction separation), this method has the advantages of large production capacity, simple process and easy to achieve continuously.
Principle
The extraction agents used for the separation of zirconium and hafnium mainly include ketone extractants, neutral phosphorus-containing extractants and amine extractants.
A commonly used ketone extractant is methyl isobutyl ketone (MIBK), which can form a neutral extract with hafnium thiocyanate and is preferentially extracted into the organic phase.
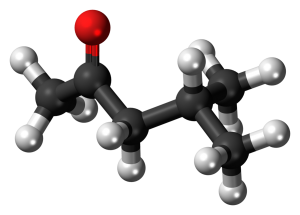
A typical neutral phosphorus-containing extractant is tributyl phosphate (TBP), which is preferentially extracted into the organic phase through the coordination of oxygen atoms in chemical bonds with zirconium metal atoms to form a neutral extract compound Zr(NO3)4•2TBP.
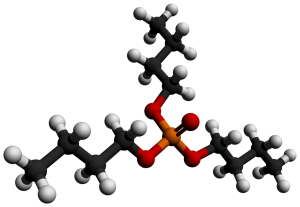
The commonly used amine extractant is trioctylamine (TOA). Trioctylamine forms an extract with zirconium ions in an acidic medium, and is preferentially extracted into the organic phase.
Process flow
There are three extraction processes: MIBK, TBP, and N235.
MIBK extraction
It uses ZrCI4 as raw material, adds water and NH4CNS ingredients. MIBK preferentially extracts hafnium, leaving a large amount of zirconium in the aqueous phase. This is the earliest extraction process used to separate zirconium and hafnium, and it is adopted by major producers of zirconium and hafnium such as the United States, France, Germany, and Japan.
TBP extraction
There are two aqueous feed systems for this process: nitric acid, and a mixed acid of nitric & hydrochloric acid. The former is to convert the product of zircon decomposed by alkali fusion method into nitric acid aqueous phase feed liquid, and use TBP to preferentially extract zirconium; the latter use Zr-CI4 as raw material, add water, nitric acid and hydrochloric acid as ingredients, and then use TBP to preferentially extract zirconium.
The separation coefficient of zirconium and hafnium in the TBP extraction process is large, and the number of extraction stages is small, and atomic-level zirconium oxide and hafnium oxide can be obtained at the same time. However, the water-phase feed liquid is highly corrosive, and the emulsification problem in the extraction process has not been completely solved, thus affecting its popularization and application.
N235 extraction
First, the zircon is decomposed by alkali fusion method, and the product is washed with water to remove silicon, and then leached with sulfuric acid to obtain a sulfuric acid solution of zirconium, and then the zirconium is preferentially extracted with N235. After washing, atomic-level zirconia containing hafnium <0.01% can be obtained. The hafnium in the raffinate is enriched to 50% to 70%, and then extracted by P204, and the zirconium and hafnium are further separated to obtain atomic energy level hafnium oxide containing more than 96% of hafnium.
This process has low material toxicity, light equipment corrosion, stable operation, and easy disposal of waste, so it is currently recognized as one of the best extraction processes.
Extraction equipment
There are two main types of extraction equipment, one is the extraction tower, and the other is the box-type mixer-settler. The former is used by the MIBK process, and the latter is used by TBP and N235 extraction process. The extraction tower occupies a small area and has a large production capacity. The box-type mixer-clarifier is simple in structure and stable in operation, and is generally made of acid-resistant materials such as plastic or plexiglass.
For more information, please visit https://www.samaterials.com/.
One thought on “Separation of Zirconium and Hafnium by Solvent Extraction”